On-Site Microfluidic Assay for the Detection of Multiple Drugs in Urine
ACS Omega 2026, 11, 8, 13697–13705: Graphical abstract
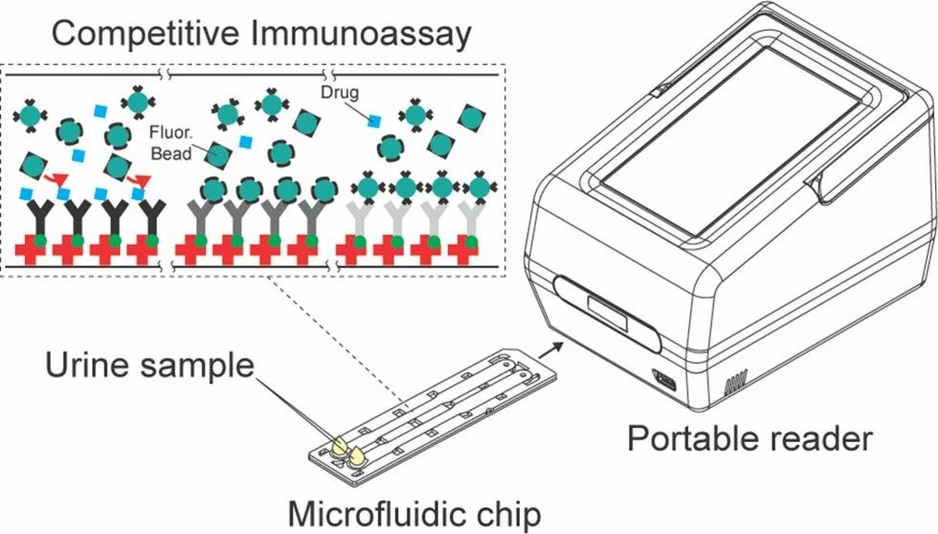
This study introduces a portable fluorescence-based microfluidic assay for rapid detection of multiple drugs in urine, including methamphetamine, benzoylecgonine, THC-COOH, and benzodiazepines. The system combines a compact reader with a dual-channel chip containing drug-specific antibodies, enabling simultaneous analysis within 5 minutes.
Validation with 141 authentic urine samples demonstrated high sensitivity, specificity, and minimal cross-reactivity for both single and multiple drug use. The assay offers a cost-effective and field-deployable alternative to conventional immunoassays, supporting on-site forensic and clinical drug screening.
The original article
On-Site Microfluidic Assay for the Detection of Multiple Drugs in Urine
Sanghag Ko, Sora An, Hyunjun Bae, Joonseok Seo, Suncheun Kim, Jeongmin Lee, Yohan Jeong, Juhyung Kim, Sangki Lee, Hyung Chul Kim, Seok Chung*, and Heesun Chung*
ACS Omega 2026, 11, 8, 13697–13705
https://doi.org/10.1021/acsomega.5c11027
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
Forensic, clinical, and workplace testing for drugs of abuse in urine typically involves immunoassays and mass spectrometry (MS)-based methods. (1,2) Immunoassays are commonly used for rapid initial screenings, while MS-based assays are reserved for confirmatory testing. (3−5)
A variety of immunoassay platforms have been developed for efficient drug screening. The lateral flow assay (LFA), a commonly used presumptive test in point-of-care test kits, offers rapid results within 5–10 min. These portable and inexpensive kits require minimal training, making them suitable for workplaces, schools, and roadside screening. (6−9) However, they are limited by relatively low sensitivity and specificity, which may lead to false positives and negatives. Therefore, positive results must be confirmed by MS-based methods. (1,2,4) On the other hand, laboratory-based immunoassays are automated analytical systems employed in forensic and clinical laboratories and utilized in workplace drug testing programs to detect drugs in biological samples with high throughput and precision. These systems commonly employ platforms such as enzyme-linked immunosorbent assay (ELISA), enzyme-multiplied immunoassay technique (EMIT), cloned enzyme donor immunoassay (CEDIA), and chemiluminescent immunoassay (CLIA). Nevertheless, their high operational cost and dependence on complex instrumentation restrict its use in point-of-care settings. (10−13)
Paper-based microfluidic assays are widely used in point-of-care diagnostics and drug testing because of their simplicity, portability and accessibility. (14,15) In forensic applications, these platforms have also been developed for the detection of drugs and their metabolites in urine, as well as for alcohol and drug identification. (16,17) Despite being both user- and manufacturer-friendly, paper-based systems still face limitations inherent to cellulose substrates, including pore size variability, nonspecific binding, and restricted fluid-control precision. (14,18)
To overcome these challenges, we developed an optimized microfluidic assay featuring nanointerstice integration, enabling rapid testing. (19−21) Unlike previous systems, our design operates entirely without external forces, instead leveraging capillary pressure to drive flow and nanointerstices to accelerate fluid filling.
The microfluidic assay in this study includes a portable reader whose size is smaller (dimensions 12.7 cm × 15.0 cm × 20.6 cm, weight 2 kg, Figure S1). Microfluidic chip in which four drug-specific antibodies are immobilized across two channels. The design is optimized to minimize errors and stabilize the flow for precise results.
To apply this assay, four target drugs were selected based on both global prevalence and recent domestic forensic trends. Globally, drug misuse has risen by approximately 26% over the past decade, with the darknet transactions being largely dominated by cannabis (48%), amphetamine-type stimulants (16%), cocaine (12%), and benzodiazepines (6%). (22) Recent UNODC and EMCDDA reports similarly emphasize the ongoing increase in methamphetamine trafficking, record-high levels of global cocaine supply, the continued predominance of cannabis use, and the rising nonmedical use and diversion of benzodiazepines. (23,24) In Korea, forensic drug case analyses in 2024 revealed that methamphetamine and cannabis were the most frequently identified substances. Notably, cocaine cases increased by 291% compared to the previous year, while benzodiazepines-related cases also showed a significant 40% year-overyear increase. (25) Considering these trends, methamphetamine, cocaine, cannabis, and benzodiazepines were selected as the primary targets of this study. For the detection of these substances in urine, antibodies specific to the parent compounds were used for methamphetamine (MET) and benzodiazepines (BZOs), while detection of cocaine and cannabis was achieved through antibodies against their primary urinary metabolites, benzoylecgonine (BE) and 11-nor-9-carboxy-tetrahydrocannabinol (THC-COOH), respectively. (26−28)
The performance of the developed microfluidic assay was validated using both drug-spiked and authentic urine specimens to ensure its reliability under both controlled and actual forensic testing environments. Drug-spiked specimens were prepared in the laboratory and utilized to optimize assay parameters such as reagent concentration, flow rate, and detection sensitivity. Following the evaluation with drug-spiked specimens, authentic drug-positive urine specimens, provided by the National Forensic Service in Korea (NFS) and Paraguay (Lab HORVATH), were used to assess the practical applicability and diagnostic accuracy of the assay under realistic forensic testing scenarios.
Therefore, the aim of this study was to develop and validate a portable microfluidic assay optimized for the simultaneous detection of four major drugs of abuse in urine─methamphetamine, cocaine, cannabis, and benzodiazepines─under realistic forensic testing conditions. By integrating nanointerstice technology and drug-specific antibodies within a compact platform, the assay was designed to address the limitations of existing point-of-care and laboratory-based immunoassay methods. Through performance evaluations using both drug-spiked and urine samples, this study was conducted to demonstrate the practical applicability, reliability, and diagnostic accuracy of the microfluidic assay for use in rapid on-site drug screening.
2. Materials and Methods
2.4. Standard Preparation and Sample Acquisition
Gas chromatography–mass spectrometry (GC-MS) was used to confirm urine specimens requiring verification. Analyses were conducted using an Agilent 7890B GC coupled to an Agilent 5977A mass selective detector (electron–ionization mode). Helium was used as the carrier gas at 1.0 mL/min, and samples (1 μL) were injected in splitless mode. Separation was achieved on an HP-5 ms capillary column (30 m × 0.25 mm × 0.25 μm) with an oven program of 80 °C (1 min), ramped at 10 °C/min to 280 °C and held for 5 min (total run time: 28 min). The inlet and transfer-line temperatures were 280 and 280 °C, respectively. Qualitative identification was performed using selected-ion monitoring (SIM) targeting characteristic ions for each analyte.
3. Results
3.1. Microfluidic Chip and Portable Reader
This study employs a microfluidic assay that incorporates a simple sample preparation method, in which the urine specimen is diluted with a sample buffer (Absology, cat.no. ASB3) at a 1:10 ratio to minimize urine matrix effects and control the viscosity of the flow in the channel. 50 μL of the mixture is introduced onto a microfluidic chip for analysis with a potable reader (Figure 1).
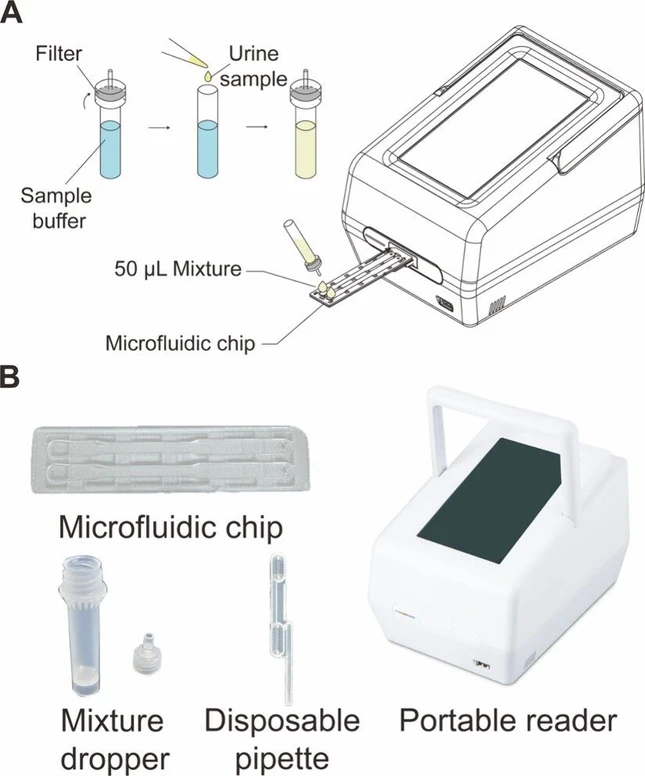 ACS Omega 2026, 11, 8, 13697–13705: Figure 1. Microfluidic assay. (A) Pretreatment of the urine specimen. To prepare the measurement, the user mixes the urine specimen with specimen buffer at a 1:10 ratio in the mixture dropper and then loads 50 μL of the mixture into each channel. (B) Photographs of the microfluidic assay. It shows the main elements of the assay system, comprising the microfluidic chip, mixture dropper, disposable pipet, and the portable reader device.
ACS Omega 2026, 11, 8, 13697–13705: Figure 1. Microfluidic assay. (A) Pretreatment of the urine specimen. To prepare the measurement, the user mixes the urine specimen with specimen buffer at a 1:10 ratio in the mixture dropper and then loads 50 μL of the mixture into each channel. (B) Photographs of the microfluidic assay. It shows the main elements of the assay system, comprising the microfluidic chip, mixture dropper, disposable pipet, and the portable reader device.
3.5. Measurement of Urine Specimens in the Microfluidic Chip
Following the standard validation, we analyzed 141 authentic urine specimens: N = 30 methamphetamine users, N = 38 cocaine users, N = 30 cannabis users, and N = 13 benzodiazepine users, while the nondrug user group included 30 drug-free urine specimens (Supporting Information 2).
The results obtained from the microfluidic assay were compared with those from the COBAS c311 for MET, THC-COOH, BZOs, and a point-of-care test kit test for BE. Figure 6A confirms no detection of MET, BE, THC-COOH, and BZOs in the nondrug user group. In Figure 6B, COI values for MET exceeded 1 in all methamphetamine specimens, with THC-COOH in one specimen, and no BE and BZOs detected. The sensitivity of the microfluidic assay─defined as the percentage of true positives─and the specificity─defined as the rate of true negatives─ (32)were both 100% (30/30) for MET when compared to COBAS c311. Figure 6C shows that COI values for BE exceeded 1 in all cocaine specimens, with THC-COOH and BZOs detected in 34 and 9 specimens, respectively. The microfluidic assay demonstrated 100% (38/38) sensitivity and 100% (30/30) specificity in the detection of BE when compared to the point-of-care test kit. In Figure 6D, all cannabis specimens had COI values above 1 for THC-COOH, and one specimen showed a COI above 1 for MET; no BE and BZOs were found in these specimens. Sensitivity and specificity for THC-COOH were both 100% (30/30) as compared to COBAS c311. Figure 6E indicates that COI values for BZOs exceeded 1 in 6 out of the 13 benzodiazepine specimens, demonstrating a lower sensitivity of 46% (6/13), though specificity remained at 100% (30/30) compared to COBAS c311. In addition to single-drug analysis, specimen-level evaluation revealed that the microfluidic assay was also capable of detecting multidrug users. As shown in Figure 6F, among the total 141 specimens, 30 were drug-free, 75 were single-drug users, and 36 were multidrug users. This demonstrates the assay’s ability to distinguish complex drug use patterns in forensic samples.
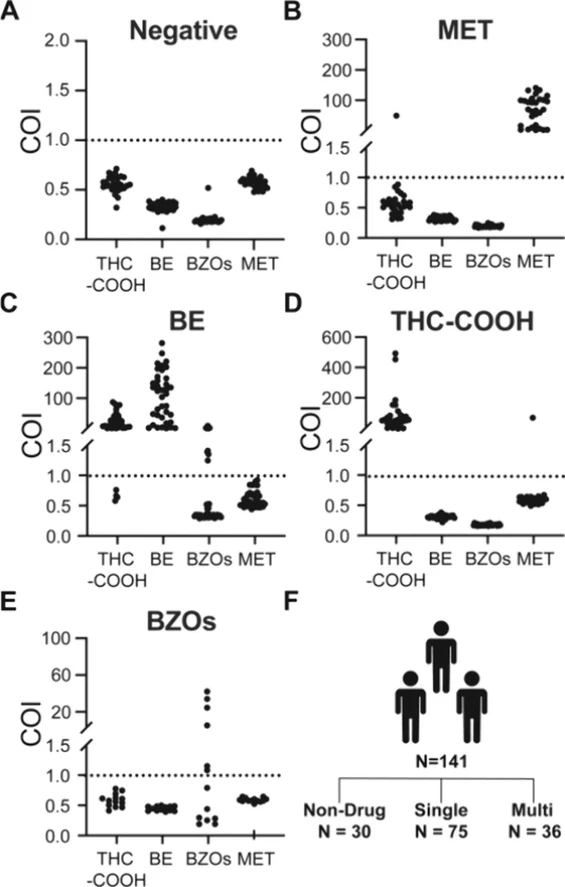 ACS Omega 2026, 11, 8, 13697–13705: Figure 6. Drug use patterns in N = 141 samples and their cutoff index (COI) results. COI results of (A) N = 30 individuals who did not consume any of the four drugs. (B) N = 30 individuals who consumed at least methamphetamine, compared with GC-MS results. (C) N = 38 individuals who consumed at least cocaine, compared with a rapid test kit. (D) N = 30 individuals who consumed at least cannabis, compared with GC-MS. (E) N = 13 individuals who consumed at least benzodiazepines, compared with LC-MS. (F) Samples consisted of nondrug group (N = 30), abusers who consumed one type of drug (N = 75), and others who took more than two types of drugs (N = 36).
ACS Omega 2026, 11, 8, 13697–13705: Figure 6. Drug use patterns in N = 141 samples and their cutoff index (COI) results. COI results of (A) N = 30 individuals who did not consume any of the four drugs. (B) N = 30 individuals who consumed at least methamphetamine, compared with GC-MS results. (C) N = 38 individuals who consumed at least cocaine, compared with a rapid test kit. (D) N = 30 individuals who consumed at least cannabis, compared with GC-MS. (E) N = 13 individuals who consumed at least benzodiazepines, compared with LC-MS. (F) Samples consisted of nondrug group (N = 30), abusers who consumed one type of drug (N = 75), and others who took more than two types of drugs (N = 36).
5. Conclusions
Immunoassay face distinct limitations. LFAs often suffer from low sensitivity and subjective result interpretation, while laboratory-based immunoassay systems require high costs and offer poor portability. In response to these challenges, this study developed a portable microfluidic assay for on-site screening. The assay enables semiquantitative detection of four drugs─methamphetamine, cocaine, cannabis, and benzodiazepines─using a microfluidic chip integrated with drug-specific antibodies. It provides clear positive/negative results, minimizing subjective interpretation, and also supports semiquantitative numerical outputs through COI values. Based on analysis of 141 authentic urine specimens tested, including 36 cases of polydrug use, the microfluidic assay demonstrated accurate and reliable detection across all four major drug classes, enabling robust performance even in polydrug use cases. The total analysis time remains under 5 min, ensuring practical usability for on-site testing. Its multiplexing capability allows for the simultaneous detection of multiple drugs in a single run, improving efficiency and utility in field testing scenarios such as roadside and workplace drug monitoring.
Given those technological advantages and demonstrated performance, this assay represents a promising tool for broader field applications. Future research may focus on expanding the assay’s applicability to a wider range of detectable drug panel, improving its analytical throughput, or integrating wireless connectivity features for real-time monitoring and cloud-based data sharing.