Menu
More information
WebinarsAbout usContact usTerms of use
LabRulez s.r.o. All rights reserved. Content available under a CC BY-SA 4.0 Attribution-ShareAlike
Active
Inactive
GCMS webinars focusing on Academy/Principles - page 13
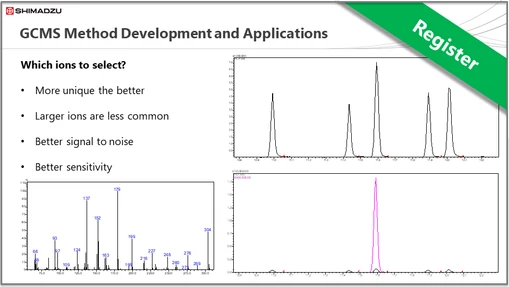
Shimadzu Theory & Key Principles Series - GCMS Session 5: GCMS Method Development and Applications
This is the fifth webinar on fundamental concepts in GCMS. This session will cover: General method set-up, Analysis aims, Column choice, Ion selection, Improving chromatography, Improving sensitivity.
RECORD
|
Already taken place We, 4.11.2020
Shimadzu Corporation


Learn How to Optimize Your GCMS Method Parameters for Success
In this webinar we will discuss many parameters that can be optimized to attain the most power from the mass spectrometer such as sampling rates, simultaneous Full Scan/SIM conditions and many more.
RECORD
|
Already taken place We, 21.10.2020
PerkinElmer


Shimadzu Theory & Key Principles Series - GCMS Session 4: GCMS Operation and Maintenance
This is the fourth webinar on fundamental concepts in GCMS. This session will cover: Consumables for GCMS, Gas filters, Column installation, Source maintenance, Leak checking, Tuning
RECORD
|
Already taken place We, 21.10.2020
Shimadzu Corporation

-GCMS_s.webp)
Shimadzu Theory & Key Principles Series - GCMS Session 3: Triple Quadrupole (TQ) GCMS
This is the third webinar on fundamental concepts in GCMS. This session will cover: Triple Quadrupole (TQ), Collision cell, Precursor & Product ions, Analysis modes, Benefits of TQ.
RECORD
|
Already taken place We, 7.10.2020
Shimadzu Corporation
-GCMS_s.webp)

MilliporeSigma - Reference Materials in Pharma QC
This webinar will provide an overview of key concepts relating to Reference Materials. Reference Material manufacture and characterization. Detail Certificate of Analysis (CoA). Reference Material quality.
RECORD
|
Already taken place We, 23.9.2020
Business Review Webinars

Related content
More information
WebinarsAbout usContact usTerms of use
LabRulez s.r.o. All rights reserved. Content available under a CC BY-SA 4.0 Attribution-ShareAlike