Is Curzerene Responsible for the Bioactive Properties of Eugenia uniflora? A Possible Misinterpretation of Bioactive Markers
ACS Omega 2025, 10, 48, 59425–59434: Graphical abstract
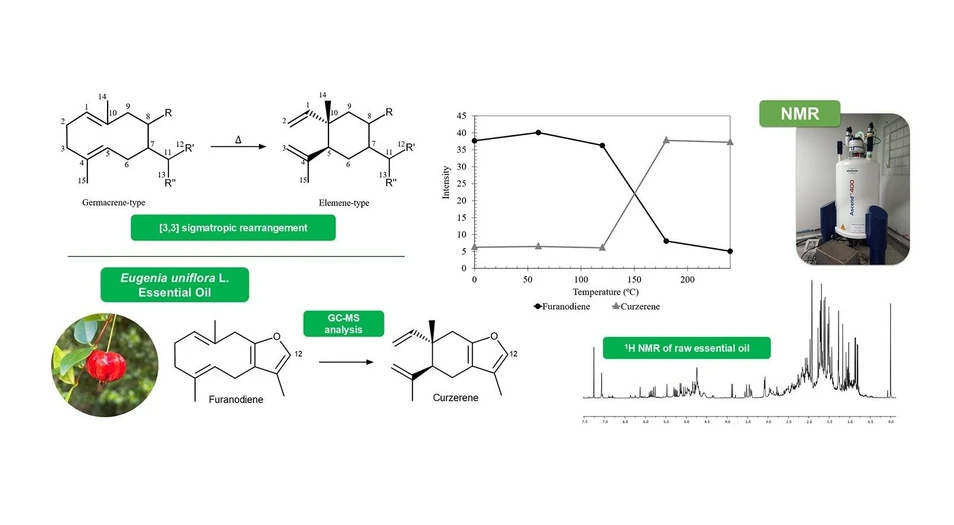
This study investigates the chemical transformations of Eugenia uniflora essential oil under thermal conditions simulating gas chromatography analysis. GC–MS and NMR revealed that sesquiterpenes of the germacrene type, such as furanodiene, undergo sigmatropic rearrangement to form elemene-type compounds, including curzerene.
Thermal treatment confirmed the formation of rearrangement products, indicating that bioactive properties historically attributed to curzerene may instead originate from furanodiene. The findings highlight the importance of combining complementary analytical techniques, such as NMR, to avoid misinterpretation of essential oil composition and bioactivity markers.
The original article
Is Curzerene Responsible for the Bioactive Properties of Eugenia uniflora? A Possible Misinterpretation of Bioactive Markers
Vinicius Monteiro Schaffka, Raphaela Pereira Guaringue, Larissa Kozan, André Luis Kerek, Cássia Gonçalves Magalhães, Andersson Barison, and Barbara Celânia Fiorin*
ACS Omega 2025, 10, 48, 59425–59434
https://doi.org/10.1021/acsomega.5c08966
licensed under CC-BY 4.0
Selected sections from the article follow. Formats and hyperlinks were adapted from the original.
Essential oils are a matrix of volatile and complex compounds characterized by strong odors and significant biological activity. In plants, they play a crucial role in defense against bacteria, viruses, and predators. (1) These biological properties support the extensive use of essential oils in the food, cosmetics, and pharmaceutical industries. (1,2) Eugenia uniflora L., commonly known as pitangueira, is a native Brazilian tree widely distributed across South America and is well-known for its bioactive essential oil. (3) Beyond the consumption of its fruits in natura or functional beverages, its leaves are used in infusions, decoctions, and tinctures to treat a variety of diseases, including diarrhea, stomach pain, worm infestations, fever, flu, hyperglycemia, hyperlipidemia, and hypertension. (4,5) Studies have shown that the composition of secondary metabolites in E. uniflora can vary depending on factors such as fruit color, ripening stage, (6) and seasonal influences. (4,7−10)
The essential oil of E. uniflora is predominantly composed of sesquiterpenes, mainly germacrene-type, such as curzerene, germacrene B, germacrone, germacrene D, selina-1,3,7(11)-trien-8-one, and its oxide. Additionally, Elemene-type terpenes are also reported, including furanodiene, β-Elemene, and β-Elemenone. (11−13)
However, those sesquiterpenes are well-documented for their thermal instability. When heat is applied, germacrene-type sesquiterpenes undergo a [3,3]-sigmatropic rearrangement, known as Cope rearrangement, converting into Elemene-type terpenes as presented in Figure 1. (14−16) This reaction is widely utilized as a synthetic pathway to produce Elemenes from germacrene precursors. (17,18)
![ACS Omega 2025, 10, 48, 59425–59434: Figure 1. [3,3]-Sigmatropic rearrangement of Germacrene-type into Elemene-type.](https://gcms.labrulez.com/labrulez-bucket-strapi-h3hsga3/ACS_Omega_2025_10_48_59425_59434_Figure_1_3_3_Sigmatropic_rearrangement_of_Germacrene_type_into_Elemene_type_250708e73c_l.webp) ACS Omega 2025, 10, 48, 59425–59434: Figure 1. [3,3]-Sigmatropic rearrangement of Germacrene-type into Elemene-type.
ACS Omega 2025, 10, 48, 59425–59434: Figure 1. [3,3]-Sigmatropic rearrangement of Germacrene-type into Elemene-type.
Gas chromatography (GC) is the main technique for characterizing essential oils, often coupled with mass spectrometry (GC-MS) and the retention index determination is used in the identification of the compounds through comparison with databases. (21) During GC analysis, essential oils are subjected to multiple heating stages. Initially, the sample is exposed to a high inlet temperature, typically around 240 °C, to ensure complete volatilization. It then enters the oven, where the temperature follows a programmed ramp starting at 60 °C, increasing at 3 °C/min, and reaching up to 240 °C. Finally, the sample encounters the elevated temperatures of the mass spectrometry (MS) detector’s source (250 °C) and interface (280 °C). Even in isocratic methods, where the oven temperature remains constant throughout the analysis, the inlet, source, and interface temperatures are maintained at high levels. Although the sample passes through these stages briefly, the elevated temperatures can induce Cope rearrangements in the essential oil, leading to possible changes in its composition. (22,23)
The accurate characterization of the chemical composition of essential oils is crucial for any study employing them as a biological or analytical matrix, as it supports the interpretation of bioactivity, chemotaxonomic classification, and quality control of samples with economic value added. There are many studies attributing relevant biological activities, classifying specimens, and identifying phenotypic and genetic variations based on the chemical composition of the essential oil of E. uniflora L., which is usually characterized by GC-MS. (8,9,11,24) As an example of the study carried out by de Jesus, found acute anti-inflammatory effect of a rich in curzerene essential oil. (5) Similarly, Pascoal, reported distinct essential oil composition patterns among E. uniflora varieties─yellow, red, and purple fruits─primarily driven by variations in β-Elemene and germacrene D content. (7) These findings highlight metabolic changes during fruit ripening and underscore the influence of genetic diversity among E. uniflora varieties. Such exhibitions are unsatisfactory, because they sustain all their evidence in the characterization of their essential oil only by GC-MS analysis, without considering the Cope rearrangements, a fact that may change the outcome of their research. In this context Nuclear Magnetic Resonance (NMR) spectroscopy is a good alternative for essential oil analysis, since is a robust not destructive technique and able to detect and quantify simultaneously a wide range of metabolites that exceed the concentration of about 5–10 μM. (25)
In order to highlight analytical concerns about the uses of only GC-MS data to access chemical relevance of E. uniflora essential oil studies, Nuclear Magnetic Resonance (NMR) spectroscopy was employed to evaluate the chemical composition of the essential oil of Eugenia uniflora L. before and after an induced thermal treatment, compared to GC-MS data. The findings provide insights into thermolabile compounds and highlight potential misidentifications of the bioactive properties associated with the sesquiterpenes of E. uniflora present in literature.
2. Results and Discussion
2.1. Characterization of the Essential Oil
Approximately 60 compounds, mostly sesquiterpenes (Germacrene B) and oxygenated sesquiterpenes (Germacrone), were visualized in the essential oil, with the 9 compounds having a relative area above 2%, as shown in Table 1. The identified and most prevalent compounds align with those reported in the literature for E. uniflora. (4,11,12) It is possible to highlight the significant proportion of Germacrene-type sesquiterpenes compared to Elemene-type, except Curzerene, which was found in high concentration.
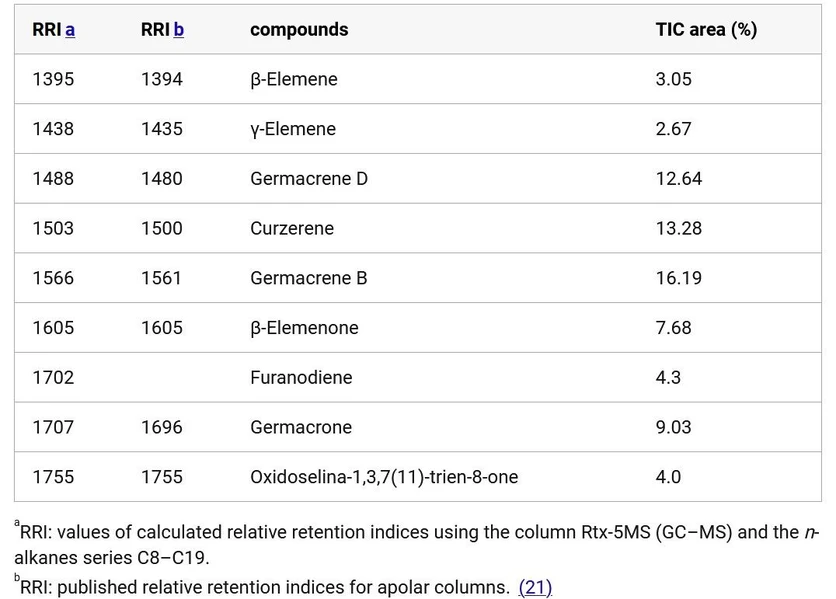 ACS Omega 2025, 10, 48, 59425–59434: Table 1. Main Components Identified in the GC–MS Analysis of the Essential Oil from Leaves of E. uniflora.
ACS Omega 2025, 10, 48, 59425–59434: Table 1. Main Components Identified in the GC–MS Analysis of the Essential Oil from Leaves of E. uniflora.
The variation in the composition of the essential oil from E. uniflora leaves was described in literature, (9) who classified the specimens into three groups: Group I, containing yellow, dark red, and purple fruits, which showed high concentrations of germacrene B, germacrone, and atractylone; Group II, consisting of light red fruit samples, with high concentrations of curzerene, germacrene D, and germacrene A; and Group III, containing orange-red fruits, with a high content of selina-1,3,7(11)-trien-8-one and oxyselina-1,3,7(11)-trien-8-one. According to this classification, this essential oil fits into Group II.
Chemical transformations of many components found in the E. uniflora leaves essential oil result from the heating conditions typically applied during GC–MS analysis, which occur through Cope Rearrangement. (12,19) Cope rearrangement is a stereospecific [3,3]-sigmatropic rearrangement that proceeds via the most stable chairlike transition state of 1,5-cyclodecadiene, from germacrene-type, to give 1,2-divinylcyclohexane, known as Elemene-type. (14) Some of the Cope rearrangements found in the E. uniflora essential oil are represented in Figure 2. (11,22,23,26)
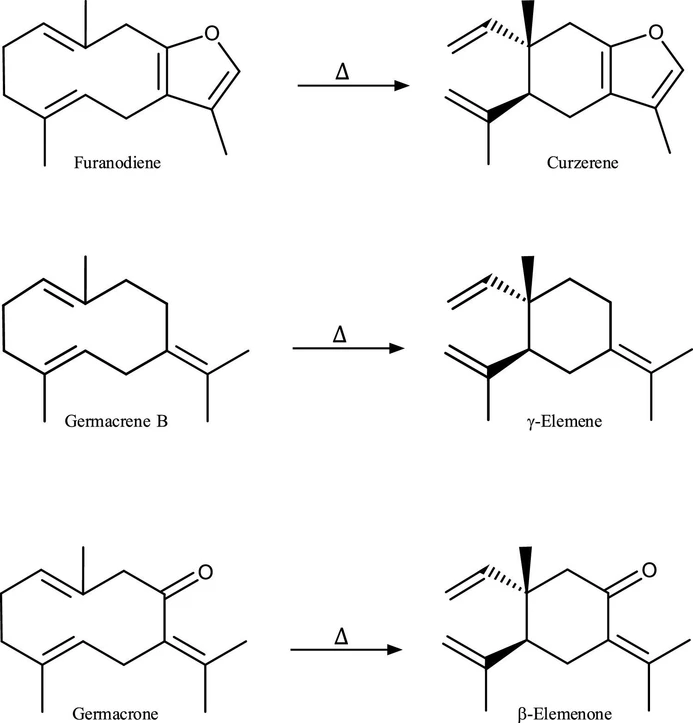 ACS Omega 2025, 10, 48, 59425–59434: Figure 2. Cope Rearrangements of sesquiterpenes.
ACS Omega 2025, 10, 48, 59425–59434: Figure 2. Cope Rearrangements of sesquiterpenes.
The same essential oil was analyzed using NMR spectroscopy to identify its components. Although NMR identification is considerably more labor-intensive than GC-MS analysis, it offers the advantage of being a cold and nondestructive technique. Since the sample does not undergo heating during the process, its stability is preserved throughout the analysis. The conformational differences observed in germacrene-type compounds further increase the spectrum’s complexity, leading to signal broadening in the 1H NMR spectra. (27)
A substantial number of signals were observed in the 1H spectrum, with the majority concentrated between 0.5 and 3.0 ppm, a region characteristic of terpene compounds. The region between 4.5 and 6.0 ppm also exhibited numerous signals attributed to vinylic protons, observed in sesquiterpenes (Figure 3).
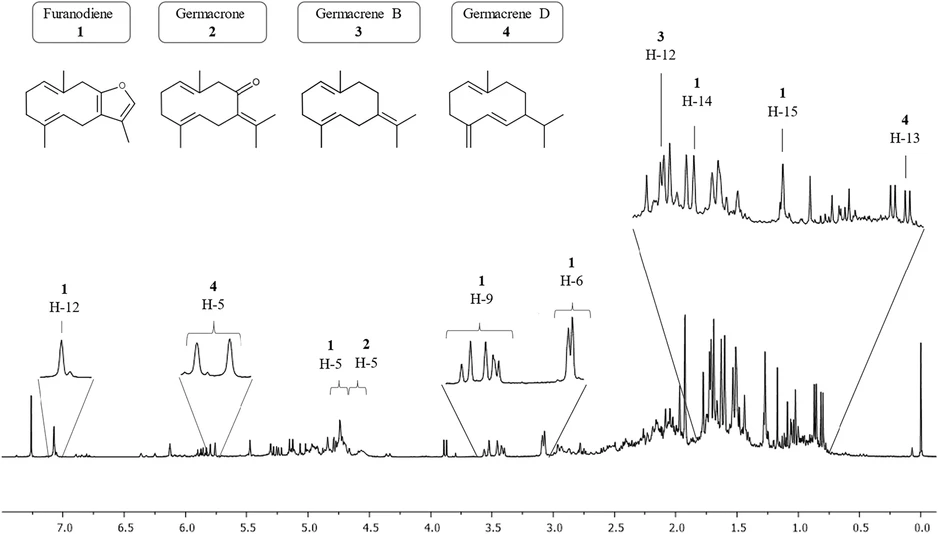 ACS Omega 2025, 10, 48, 59425–59434: Figure 3. 1H NMR (400.13 MHz, CDCl3) spectra of the raw essential oil.
ACS Omega 2025, 10, 48, 59425–59434: Figure 3. 1H NMR (400.13 MHz, CDCl3) spectra of the raw essential oil.
3. Conclusions
The investigation of the composition of E. uniflora essential oil by NMR and the thermal treatment performed proved that curzerene may come from furanodiene due to the transformations that occur in the matrix during gas chromatographic analysis. These findings have significant implications for the investigation into the chemical composition of E. uniflora essential oil and the evaluation of the biological properties of its sesquiterpenes. Since each technique has advantages and disadvantages, combining GC-MS analysis with a cold technique, such as NMR is important to guarantee the proper identification of the compound genuinely responsible for any property assigned to E. uniflora essential oil.
4. Materials and Methods
4.2. GC-MS Analysis
The gas chromatographic analyses of the essential oils (10 mg mL–1) were performed using a gas chromatography–mass spectrometry system (Shimadzu GCMS-QP2020 Gas Chromatograph). The analyses were conducted on an RTx-5MS capillary column (30 m × 0.25 mm internal diameter × 0.25 μm film thickness), and the analytical conditions were as follows: split ratio of 1/10, injector temperature at 250 °C, ion source at 250 °C, and interface at 280 °C. The temperature program of the oven was set at 60 °C for 5 min, followed by a temperature ramp of 3 °C/min until reaching the final temperature of 240 °C. The components were identified based on the relative retention index, calculated for each constituent by injecting a series of n-alkane standards (C8–C20) under the same sample conditions and comparing them with tabulated values as well as by comparing the obtained mass spectra with the mass spectra database and literature comparisons. (21)
4.4. NMR Analysis
All NMR spectra were acquired at 298 K on a Bruker AVANCE III spectrometer operating at 9.4 T, observing 1H at 400.13 MHz and 13C at 100.13 MHz, equipped with a broadband probe. The essential oil was solubilized in CDCl3 with 0.05% of tetramethylsilane (TMS) used as an internal reference. The 1H NMR spectra were recorded utilizing 65k time-domain data points across a spectral width of 20.00 ppm. This setup provided a digital resolution of 0.24 Hz. The data were obtained using a single 30° excitation pulse, with a relaxation delay of 1.0 s, and an average of 128 scans. The 13C NMR spectra was carried out using the zgpg30 pulse sequence utilizing 64k time-domain data points across a spectral width of 238 ppm, with an acquisition time of 1.3631 s, providing a digital resolution of 0.7434 Hz and a relaxation delay of 2.0s and an average of 2k scans.
The HSQC and HMBC experiments were conducted using standard pulse sequences from the Bruker library, with modifications to the number of acquired points to improve resolution, 2048 in F2 and 512 in F1.