Fitness for the intended use of analytical equipment and systems
Eurachem: Fitness for the intended use of analytical equipment and systems
The leaflet outlines the role and handling of analytical equipment and systems in the process of a laboratory dealing with the analysis of chemical elements and molecules. It is an introduction to topics being considered by the Eurachem Analytical Equipment and Systems Qualification Working Group.
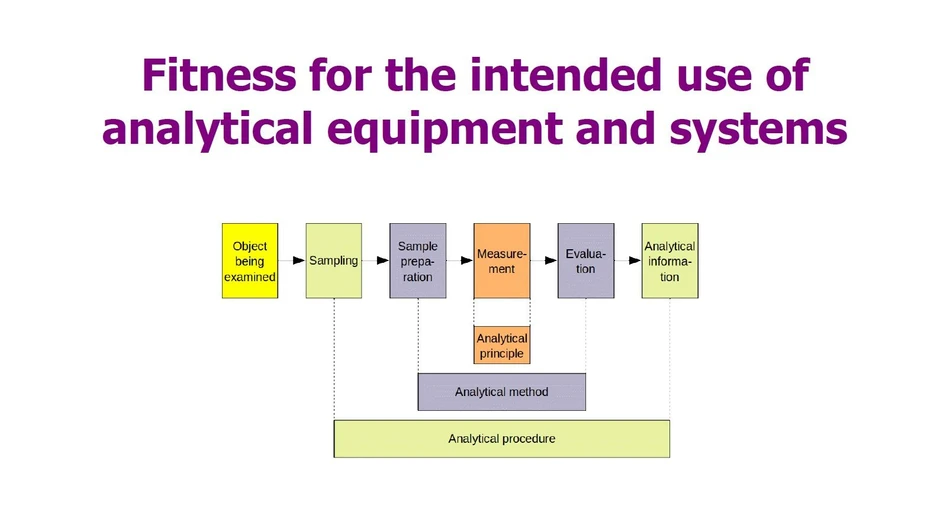
Various steps are required to draw a conclusion concerning the object being examined (sampling target), see Fig. 1. The core part, the analytical measurement, is based on an analytical principle. The measurement is based on a physical or chemical interaction, e.g. between light of a certain wavelength and the sample, which leads to a displayed measured value. This measurement is carried out using analytical equipment. This equipment must be fit for its intended use. Complex analytical equipment with extensive data processing is referred to as a system. Analytical equipment is used to perform the analytical method. The method must be validated to demonstrate that it is fit for purpose.
![Eurachem: Fig. 1 Analytical principle, method and procedure [1]](https://gcms.labrulez.com/labrulez-bucket-strapi-h3hsga3/Eurachem_Fig_1_Analytical_principle_method_and_procedure_1_b751d25236_l.webp) Eurachem: Fig. 1 Analytical principle, method and procedure [1]
Eurachem: Fig. 1 Analytical principle, method and procedure [1]
The pyramid of quality
The pyramid of quality (Fig. 2) covers the analytical method. In order to produce a reproducible, reportable result that is suitable for the intended purpose, the equipment (system) (1st level) and analytical method (2nd level) must be well defined and under control, qualified or validated, respectively.
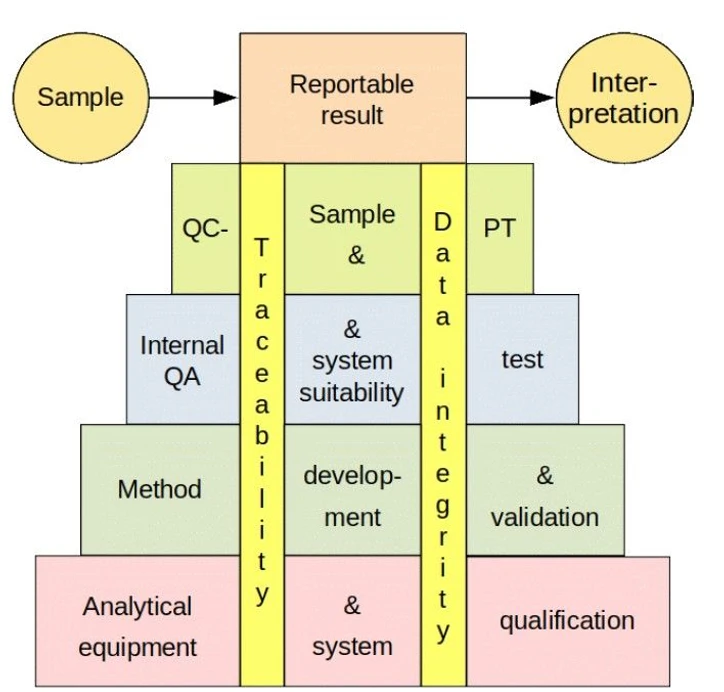 Eurachem: Fig. 2 Pyramid of quality
Eurachem: Fig. 2 Pyramid of quality
The equipment must have similar performance when carrying out the analyses as when developing and validating the method. Particularly in the case of methods based on libraries or training datasets, the long-term stability of the equipment must be ensured. In the case of transfer of the method to other equipment, both sets of equipment must have similar performance.
The performance of the equipment and the behaviour of the method are monitored by quality control measures. This is the purpose of the internal or short term (3rd level) and inter-laboratory or long term (4th level) quality assurance. When selecting the quality assurance elements, the findings from levels 1 and 2 are taken into account.
Is "calibration" sufficient?
All relevant quality assurance systems, e.g. those meeting the requirements of ISO/IEC 17025 [2] and related standards, require equipment and systems to be traceably calibrated. But is that enough to prove the fitness for the intended use of the analytical equipment?
According to the definition in VIM [3] calibration is the operation that, under specified conditions, establishes a relation between the quantity values with measurement uncertainties provided by measurement standards and corresponding indications with associated measurement uncertainties. It is implicitly assumed that the influencing quantities (e.g. ambient conditions, temperature, flow rates, intensities of signal sources, etc.) are constant and do not exhibit any short or long-term fluctuations or drifts. This assumes that no changes occur due to influencing quantities.
Nevertheless this is not the case in everyday analytical work. The influence of fluctuations or changes in the influencing quantities is evaluated through robustness investigations during method development and validation. The magnitude of the fluctuations or deviations of the equipment is specified and verified during equipment qualification.
The Fitness for Intended Use
The life cycle (Fig. 3) of new equipment to be purchased by the laboratory begins with the definition of the requirements and the intended use of the equipment.
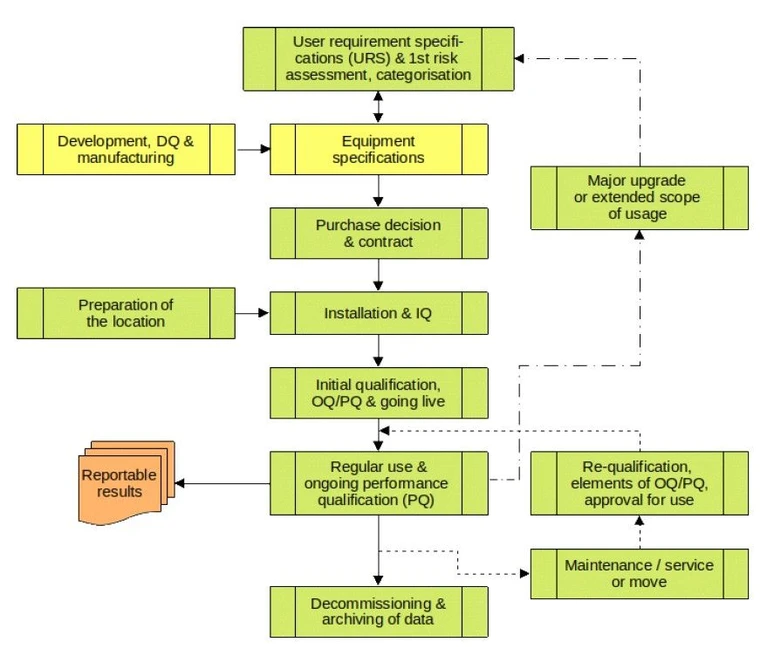 Eurachem: Fig. 3 Equipment or system lifecycle
Eurachem: Fig. 3 Equipment or system lifecycle
The User Requirement Specifications (URS) define the selection criteria and the operational ranges of the equipment parameters (influence variables), e.g. flows, temperatures, analysis volumes, etc. Considerations to support data integrity and traceability are also included in the URS. For more complex systems, the interfaces and compatibility of the components are essential.
All considerations made in this phase have an impact on the entire life cycle, i.e. right through to decommissioning. The stability and variability of the influence variables are monitored over the entire service life.
In most cases, data and metadata should remain accessible and analysable beyond the end of the equipment's life. The laboratory must use its resources as sparingly and sustainably as possible. This includes the expected service life and the costs incurred. Data integrity considerations (ALCOA+ = Attributable, Legible, Contemporaneous, Original, Accurate, + Complete, Consistent, Enduring, Available) play a role during operation and at the end of life. These include paper records and electronic records.
In the pharmaceutical industry, qualification of equipment and thus ensuring that analytical equipment and systems are suitable for the intended use, is mandatory, and the respective processes are often clearly defined. Specifications are also comprehensively defined for various equipment classes.
The ISO standards, in particular ISO 9001 [4], ISO 10012 [5], ISO/IEC 17025 [2], ISO 17034 [6] or ISO/IEC 17043 [7], cover a very large area and a wide range of sectors. For specific sectors, an interpretation of the standards is required. Eurachem guides cover requirements for analytical chemistry and related disciplines. The Analytical Equipment and Systems Qualification Working Group is therefore developing guidance on establishing the fitness for intended use of analytical equipment and systems aimed at these disciplines.
[1] Georg Schwedt, The essential guide to analytical chemistry, page 7, 1997. ISBN: 978-0-471-97412-3. Copyright Wiley-VCH GmbH and affiliated companies. All rights reserved, including those for text and data mining and training of artificial intelligence or similar technologies.
[2] ISO/IEC 17025:2017 General requirements for the competence of testing and calibration laboratories
[3] JCGM 200:2012 International vocabulary of metrology – Basic and general concepts and associated terms (VIM), 3rd edition
[4] ISO 9001:2015 Quality management systems – Requirements
[5] ISO 10012:2003 Measurement management systems - Requirements for measurement processes and measuring equipment
[6] ISO 17034:2016 General requirements for the competence of reference material producers
[7] ISO 17043:2023 Conformity assessment — General requirements for the competence of proficiency testing providers