You talk, we understand – The way out of the tower of Babel

Eurachem: You talk, we understand – The way out of the tower of Babel
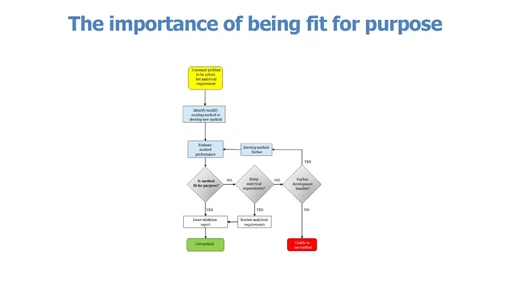
The problem
We live in a “global village” but we are all different and we use many different languages to communicate.
Even when people speak the same language, the same words may be used with different meanings. See, for example, some different uses of the word “standard” in English – a normative document, a solution of known concentration or even a type of flag.
Even in the same area of activity misunderstandings do occur which may result in a waste of time and money, or even worse consequences.
When talking about laboratory tests, lack of a full understanding of key terms and inappropriate translations leaves room for different interpretations of stated requirements. For example, there have historically been a number of different but related uses of the term ‘detection limit’. This may lead to inadequate fulfilment of those requirements, thus becoming an obstacle to the production of analytical data that is fit for its intended use. A shared interpretation of concepts related to quality in measurement between laboratories and accreditation bodies is also crucial for a fair and harmonised approach to laboratory assessment across the world.
A common language
We need a common language; clear and unambiguous. We need a common vocabulary to provide consistent definitions of concepts with their associated terms. To this end, several organisations participate in a joint effort to develop and maintain an international vocabulary of metrology (VIM) [1], which aims to achieve a unique understanding of concepts related to measurements across all sectors. The VIM is a normative reference in the standards ISO/IEC 17025, ISO 15189 and ISO/IEC 17043, and is therefore a key document for all organisations seeking accreditation.
 Eurachem: You talk, we understand – The way out of the tower of Babel_A common language
Eurachem: You talk, we understand – The way out of the tower of Babel_A common language
Is this enough?
For laboratory staff some problems still stand and need to be addressed:
- Many people are confused about both concepts and terms.
- It may be difficult to understand the “formal” VIM definitions – they are short statements intended to apply to many different measurement sectors.
- Translations into local languages may add to the confusion, e.g. if different terms are used in different sectors for the same VIM concept and the translator overlooks this issue.
- Some substantial changes to terminology have been made in an attempt to accommodate chemical and biological measurements. Some “old” concepts still stand but have new names, e.g. “within laboratory reproducibility” vs. “intermediate precision”.
To address these issues Eurachem has produced a guide [2] which provides an accessible reference containing simple and in context explanations of terms most relevant to analysts, with additional examples relating those concepts to chemical and biological measurements.
Do you understand the meaning of these terms?
Are you interested in a quantity or a quality? Depending on the answer, will you use a measurement procedure or an examination? Is method verification the same or less demanding than method validation? Do you need a calibration or just performance verification? Do you have a measurement standard in your laboratory? Is metrological traceability something you worry about? These are examples of the terms supporting the reliability of any measurement result that need to be clearly understood.
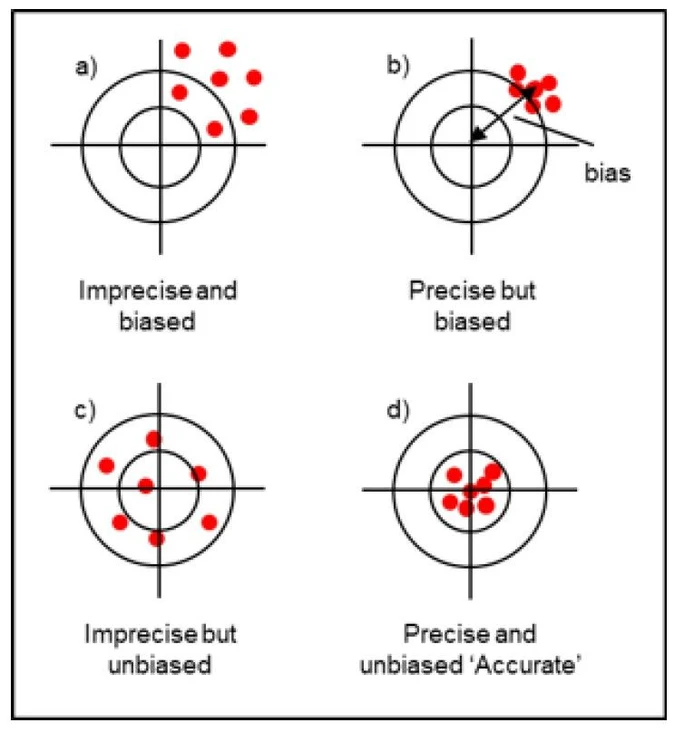
Example – Accuracy, trueness, precision: are they synonyms?
 Eurachem: You talk, we understand – The way out of the tower of Babel_Example – Accuracy, trueness, precision
Eurachem: You talk, we understand – The way out of the tower of Babel_Example – Accuracy, trueness, precision
- Closeness between measurement results (relating to the concept of precision)
- Closeness of the average of the measurement results to a reference value (relating to the concept of trueness)
- Closeness of an individual measurement result to the true value (relating to the concept of accuracy)
The different situations in the figure show the effect of:
- poor precision and trueness (imprecise and biased)
- good precision, poor trueness (precise but biased)
- poor precision, good trueness (imprecise but unbiased)
- good precision and trueness (precise and unbiased)
Improving both precision and trueness is expected to improve accuracy.
Is all this relevant to you?
Do you work in a laboratory, either as an analyst, quality manager or director? Are you involved in other activities related to analytical work, such as providing proficiency tests, producing reference materials or accrediting any such activity? Do you teach or study subjects related to measurements? Do you use measurement results or prescribe how to use them? If the answer is yes to any of these questions, the following reference documents are also for you.
[1] International vocabulary of metrology – Basic and general concepts and associated terms (VIM) 3rd edition. JCGM 200:2012. Available here. Html format with informative annotations available here.
[2] V. J. Barwick (Ed.), Eurachem Guide: Terminology in Analytical Measurement − Introduction to VIM3, (2nd ed. 2023). ISBN 978-0-948926-40-2. Available from Eurachem.