Development of potent inhibitors to boost the immune system's ability to fight cancer
IOCB Prague: Development of potent inhibitors to boost the immune system's ability to fight cancer
Our immune system is incredibly powerful: among other things, it can recognize and destroy cancer cells. But tumors can be just as effective at avoiding detection – they build “invisibility shields” to protect themselves from attack.
One breakthrough in cancer therapy has been immune checkpoint inhibitors (ICIs), drugs that reactivate immune T cells and help them fight tumors again. However, only about 20–50% of patients respond well to this treatment. Researchers are therefore seeking alternative therapeutic strategies. A small and well-known molecule called adenosine plays a key role in these efforts.
When tumor cells are stressed or dying, they release ATP, which is normally a signal that something is wrong in the tissue. But in the tumor microenvironment, ATP gets rapidly broken down by two enzymes, CD39 and CD73. Released adenosine acts like a chemical “calm-down” signal, binds to immune cells (T cells, NK cells, and others), and tells them to slow down, reducing their ability to kill cancer cells. Targeting CD73 therefore represents a promising strategy for developing new cancer treatments.
 IOCB Prague: Development of potent inhibitors to boost the immune system's ability to fight cancer
IOCB Prague: Development of potent inhibitors to boost the immune system's ability to fight cancer
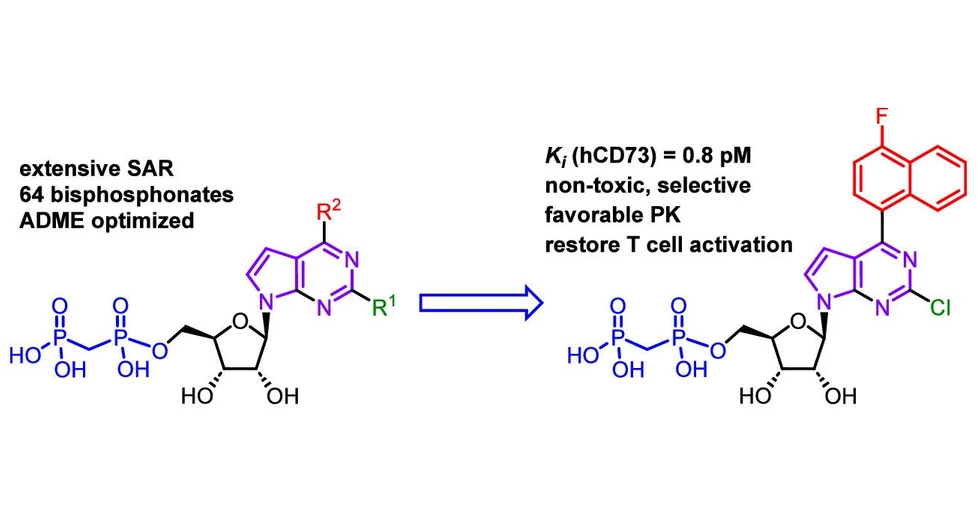
At IOCB Prague, interdisciplinary teams led by Jan Řezáč, Jan Konvalinka, and Michal Hocek used a newly developed high-throughput screening method to test over 5,000 compounds, identifying two ribonucleoside derivatives as promising CD73 inhibitors.
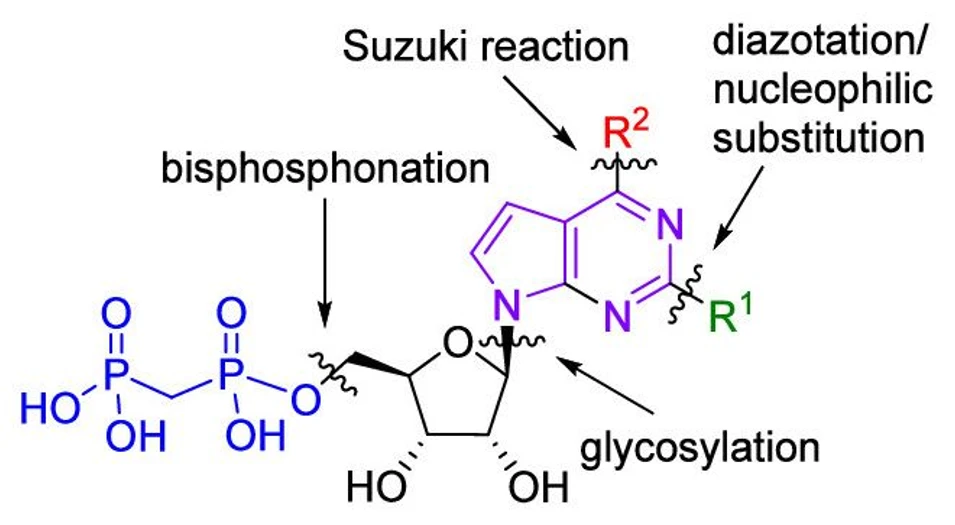
The scientists then improved the compounds’ activity by carefully modifying the ribonucleoside scaffold with bulky (het)aryl groups. Further optimization improved the pharmacokinetic properties of these compounds, yielding inhibitors with low clearance, long half-life, high solubility, and strong selectivity over related enzymes such as CD39.
In cellular experiments, the compounds reduced adenosine production in cancer cells, restored T cell activation, and showed no toxicity toward human fibroblasts. The most active compound showed a single-digit picomolar inhibition constant (Ki).
These findings establish a strong foundation for further in vivo efficacy and combination studies, which will explore the therapeutic potential of this novel inhibitor class.
The Original article
Potent Competitive Inhibitors of Ecto-5′-nucleotidase (CD73) based on 6-(Het)aryl-7-deazapurine Ribonucleoside 5′-O-Bisphosphonates
Ugnė Šinkevičiu̅tė, Magdalena Šímová, Radek Staník, Lenka Poštová Slavětínská, Kristýna Blažková, Pavel Šácha, Martin Lepšík, Jan Řezáč, Jan Konvalinka, Tereza Ormsby, Michal Tichý, Michal Hocek
ACS Pharmacol. Transl. Sci. 2026, 9, 1, 191–213
https://pubs.acs.org/doi/10.1021/acsptsci.5c00707
licensed under CC-BY 4.0
Abstract
CD73 generates immunosuppressive adenosine in the tumor microenvironment and is a promising target for cancer immunotherapy. We have designed and systematically studied diverse 2-substituted 7-deazapurine ribonucleoside 5′-O-bisphosphonates bearing a variety of (het)aryl groups at position 6 and discovered their highly potent and selective CD73 inhibition activity. The most active compounds (with single-digit picomolar Ki) contained bicyclic (het)aryl groups at position 6 in combination with chlorine at position 2. Further optimization of pharmacokinetic properties identified inhibitors with low clearance, long half-life, high solubility, and excellent selectivity over CD39 and NTPDase3. They effectively suppressed adenosine formation in MDA-MB-231 cells, rescued CD8+ T cell activation, and were nontoxic to human fibroblasts. Overall, their profile compares favorably with AB680, a CD73 inhibitor currently in phase I/II clinical trials.