Decoupling Acquisition From Data Review: Unlocking Flexibility and Efficiency in Chromatography and Mass Spectrometry Workflows

Thermo Fisher Scientific: Decoupling Acquisition From Data Review: Unlocking Flexibility and Efficiency in Chromatography and Mass Spectrometry Workflows
In a recent blog post we discussed how centralized data management enhances security and control in analytical laboratories. But beyond security, networked environments offer other architectural advantages that fundamentally change how data flows through the lab.
One key benefit is the separation of data acquisition from processing and review—a structural shift that brings measurable improvements in performance, uptime, and compliance.
 Thermo Fisher Scientific Decoupling Acquisition From Data Review Unlocking Flexibility and Efficiency in Chromatography and Mass Spectrometry Workflows
Thermo Fisher Scientific Decoupling Acquisition From Data Review Unlocking Flexibility and Efficiency in Chromatography and Mass Spectrometry Workflows
Maximizing instrument uptime
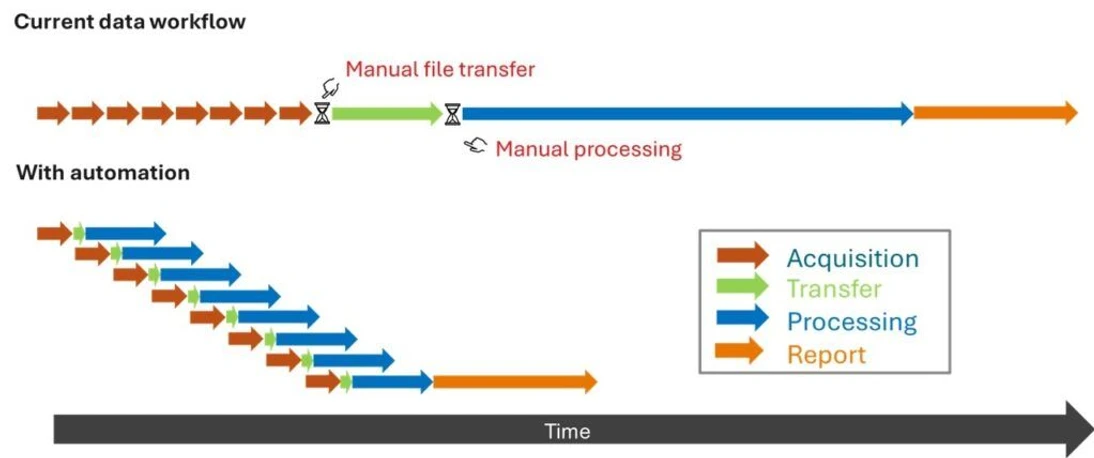
In many labs, data acquisition and review occur on the same computer, which tends to be placed next to the instrument. This ties up the acquisition workstation long after a run is complete, especially when processing tasks—such as baseline correction, peak integration, or spectral deconvolution—are resource-intensive.
 Thermo Fisher Scientific Decoupling Acquisition From Data Review Unlocking Flexibility and Efficiency in Chromatography and Mass Spectrometry Workflows
Thermo Fisher Scientific Decoupling Acquisition From Data Review Unlocking Flexibility and Efficiency in Chromatography and Mass Spectrometry Workflows
Separating these tasks means the acquisition PC can be dedicated solely to capturing raw data, while automated transfer and processing occur on a separate system. If processing is the biggest bottleneck in your workflow, this has clear advantages.
Enabling remote review and flexible access
When data is stored centrally and accessed over the network, analysts can log in from remote locations or satellite labs to perform review and approval workflows.
Central data storage also moves away from flat file sharing into a secure, user-controlled, archived storage location, which offers enhanced security and controlled access to archived data
Decoupling supports hybrid work environments and multi-user labs while supporting traceability through system audit trails and role-based access control.
Scaling processing power independently
Mass spectrometry data analysis—especially when using techniques like data-independent acquisition (DIA), MSⁿ workflows, or top-down proteomics—demands significant computational resources. These tasks benefit from multithreading, GPU acceleration, or distributed computing environments.
By shifting processing to centralized servers or virtual machines, labs can scale compute resources as needed without overloading acquisition systems, risking data loss or even sample loss from crashes during data collection.
Supporting collaborative review and shared methods
Decoupled environments allow multiple users to access and process the same dataset simultaneously, without file duplication or manual data transfer. Analysts can apply shared processing templates, compare runs side-by-side, and annotate results in a common workspace.
This is especially valuable for QA/QC teams performing multi-batch comparisons, method validation, or deviation investigations.
Enhancing compliance and traceability.
From a compliance standpoint, separating acquisition and processing improves data governance. Centralized environments can enforce version control, restrict unauthorized edits, and capture complete audit trails for every action taken—critical for GxP labs and 21 CFR Part 11 compliance. Review workflows can also be digitally signed and locked, ensuring that once approved, the data and metadata are still immutable
Conclusion
Decoupling acquisition from processing is a foundational change in how modern analytical labs operate. It reduces bottlenecks, increases system uptime, availability and enables advanced processing workflows to run on purpose-built infrastructure—without compromising compliance or productivity.
You can see how Thermo Fisher Scientific is solving these challenges in Proteomics software solution.
Visit us on LinkedIn: #Ardia #ChromatographyDataSystemSoftware, #DataManagement
Chris Knowles
Chris Knowles began his career with a PhD in mammalian cell line engineering and over a decade as a Principal Scientist and applications specialist in biopharma and bioprocessing. With deep expertise in software solutions for LC-MS, MS/MS, and advanced mass spectrometry techniques, he focuses on biologics production and analytics, including emerging areas like gene therapy, oligonucleotide therapeutics, and viral vectors. Now, as a Product Marketing Manager for BioPharma Software at Thermo Fisher Scientific, Chris is dedicated to helping customers overcome analytical challenges by delivering powerful, compliant, and user-friendly software solutions.